Short Notes: Is Matter Around Us Pure
Pure Substances
A pure substance is a material composed of only one type of particle (atom or molecule) and has a fixed composition and properties. It cannot be separated into other kinds of matter by any physical process. Elements and compounds are examples of pure substances.
Mixtures Mixtures are made up of two or more elements or compounds mixed in any ratio/proportion.
Properties
- It may be homogeneous or heterogeneous.
- The properties of constituent substances are retained.
- No new compound is formed after mixing.
- Constituents of a mixture can be separated by simple physical processes.
- It does not have a fixed melting and boiling point.

Try yourself:Mixture can be
- A.Homogeneous
- B.Heterogeneous
- C.Both (a) and (b)
- D.Pure substance
Solution
A solution is a homogeneous mixture of two or more substances (e.g., Lemonade, soda water). It has two main components: the solute (the substance that gets dissolved) and the solvent (the substance that dissolves the solute).
| Feature | Solute | Solvent |
|---|---|---|
| Definition | The substance that is dissolved in a solution | The substance that dissolves the solute |
| Quantity | Usually present in smaller amounts | Usually present in larger amounts |
| Example | Salt or sugar in water | Water (when salt or sugar is dissolved in it) |
| State | Can be solid, liquid, or gas | Can be liquid, but also sometimes a gas or a solid |

Properties of a Solution
- The particles of a solution are smaller than 1 nm (10-9 metres) in diameter. So, they cannot be seen with the naked eye.
- Because of their very small particle size, they do not scatter light passing through the solution. So, the path of light is not visible in a solution.
- The solute particles cannot be separated from the mixture by the process of filtration. The solute particles do not settle down when left undisturbed; that is, a solution is stable.
Concentration of Solution
- The amount of solute that has dissolved in a specific amount of solvent or solution is measured as solution concentration.
- A concentrated solution is one that has a significant amount of dissolved solute in it.
- A diluted solution is one that has a small amount of dissolved solute in it.

AlloysAlloys are homogeneous mixtures of metals or a mixture of a metal and another element that cannot be separated into its components by physical methods.
Examples:
- Steel – a combination of iron (metal) and carbon (non-metal).
- Bronze – a combination of copper (metal) and tin (metal).
- Brass – a mixture of copper (metal) and zinc (metal).
Suspension
A suspension is a heterogeneous mixture in which the solute particles do not dissolve but remain suspended throughout the bulk of the medium. Ex: Chalk in water, smoke in the air
Properties of Suspension :
- It is a heterogeneous mixture.
- Particles of a suspension are visible to the naked eye.
- The size of the particles is greater than 1000 nm.
- It is an unstable mixture. The solute settles down over a period of time.
- If the solution is passed through filter paper, the solute and solvent get separated.
- It scatters light when light is passed through the solution, i.e. it shows the Tyndall effect.
 Types of Solution based on particle size
Types of Solution based on particle size
Colloidal Solution
A colloid solution is a heterogeneous mixture in which the size of particles lies between that of true solutions and suspensions.
- Colloidal particles can easily scatter a beam of visible light.
- This phenomenon is called the Tyndall effect.
 Tyndall Effect
Tyndall Effect
Properties of Colloidal Solution:
- The particles of colloids can’t be seen by the naked eye individually.
- It is a heterogeneous mixture and thus the solute and solvent can’t be separated by filter paper.
- The size of particles is smaller than suspensions but greater than solutions (1 nm to 100 nm).
- It is a stable mixture. Particles do not settle down at the bottom over a period of time.
Try yourself:In sugar solution,
- A.Sugar is solute, water is solvent
- B.Sugar is solvent, water is solute
- C.Both are solutes
- D.Both are solvents.

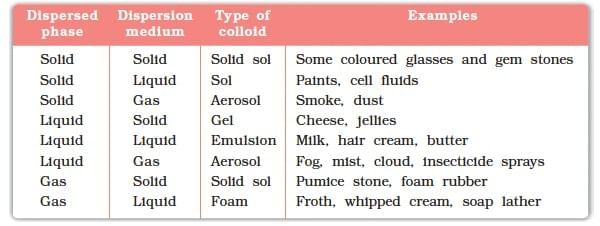 Common Examples of Colloids
Common Examples of Colloids
Physical and Chemical Change

Types of Pure Substances
The pure substance is divided into two types based on their chemical composition:
(i) ElementsAccording to Antoine Laurent Lavoisier, an element is a basic form of matter that cannot be broken down into simpler substances by chemical reactions. It is divided into three types, which are metals, non-metals and metalloids.
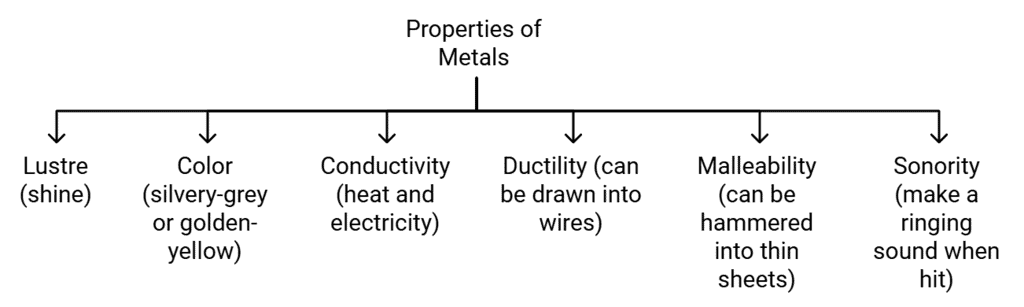 Properties of Metals
Properties of Metals
Examples: Gold, Silver, Copper, Iron, Sodium, Potassium etc. are Metals
Note: Mercury is the only metal that is liquid at room temperature.
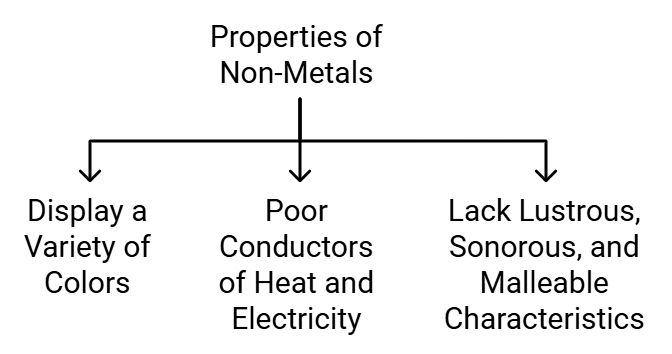 Properties of Non-Metals
Properties of Non-Metals
Examples of non-metals are hydrogen, oxygen, iodine, carbon (coal, coke), bromine, chlorine etc.
Metalloids: Elements having intermediate properties between those of metals and non-metals are called metalloids. Examples are boron, silicon, germanium etc.
Try yourself:
What is a solution?
- A.A mixture of two or more elements or compounds mixed in any ratio/proportion.
- B.A homogeneous mixture of two or more substances.
- C.A chemical reaction that forms a new compound.
- D.A mixture with a fixed melting and boiling point.
(ii) CompoundsA compound is a substance composed of two or more elements, chemically combined with one another in a fixed proportion.
Characteristics
- The properties of a compound differ from those of its constituents.
- The compound has a fixed melting point and boiling point.
- A compound is a homogeneous substance.
- Constituent elements can be separated by chemical processes.
Difference between Compounds and Mixtures

Summary
